Eye Health,Uncategorized,What's New
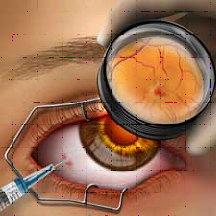 Researchers at Harvard University’s Beth Israel Deaconess Medical Center (BIDMC) and the University of New Mexico in the US are experimenting with a new eye drop treatment called Vasotide. This drug is expected to treat retinal conditions, such as age-related macular degeneration (AMD) and retinopathy of prematurity (ROP). Early results show Vasotide reduces inflammation and branching of new blood vessels in animal models of wet AMD and ROP, and this exciting new research could soon move to human trials.
Researchers at Harvard University’s Beth Israel Deaconess Medical Center (BIDMC) and the University of New Mexico in the US are experimenting with a new eye drop treatment called Vasotide. This drug is expected to treat retinal conditions, such as age-related macular degeneration (AMD) and retinopathy of prematurity (ROP). Early results show Vasotide reduces inflammation and branching of new blood vessels in animal models of wet AMD and ROP, and this exciting new research could soon move to human trials.
Current treatments for such retinal disorders are delivered as injections into the eye, which are quite expensive and can cause some patients a tremendous amount of discomfort. Vasotide is currently administered as an eye drop and as a subcutaneous injection in the animal trials; surprisingly, early results show that the Vasotide drops are as equally effective as the injections. Researchers also plan on administering the drug as self-dissolving contact lenses during the human trials. If the human trials of the drug are successful, researchers hope that the various non-injection Vasotide formulations will replace repetitive intra-ocular injections, reducing their cost and the uncomfortable experience for patients. The findings of this study thus far are published in Science Transitional Medicine.
